|
In the reactivity series, the most reactive metal is placed at the top whereas the least reactive metal is placed at the bottom. If metal A displaces metal B from its solution, it is more reactive than B. 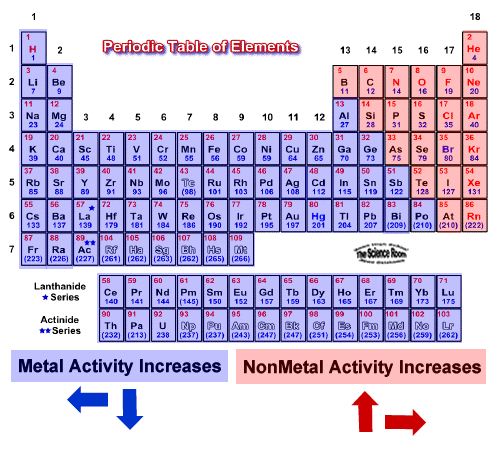
Reaction of Metals with Salt Solutions: Reactive metals can displace any metal less reactive than itself, from the oxide, chloride or sulphate of the less reactive metal in solution or their molten state. Which is most reactive group of nonmetals. Which best describes Group III B through 12 II B of the periodic table. 
Which element is a member of halogen family. Reaction of Metals with Acids: Metals are usually displace hydrogen from dilute acids (Except copper, silver and gold do not displace hydrogen from dilutes acid because they are less reactive than other metals). Which best describes a row of elements in the periodic table. They also become more reactive in water, as their reactivity increases. The alkali metals, found in group 1 of the periodic table (formally known as group IA), are so reactive that they are generally found in nature combined with other elements. They may be usually located in salts, where you can physique-focused cubic composition. These metals are most reactive when in contact withwater and air, or oil. Reaction of Metals with Water: Metals react with water to form a metal hydroxide and Hydrogen gas. The name alkali metals arises from the Arabic term al-qali, meaning ashes. Reaction of Metals with Oxygen: When metal are burnt in air, they react with the oxygen to form metal oxide. The chemical metal chemical properties of metals are discussed below: The electropositive nature allows metals to form compounds with other elements easily. Sodium metal forms sodium ions Na+, Mg forms positively charged Magnesium ions Mg2+and aluminium forms aluminium ions Al3+. Metals are very reactive and tend to losses electrons easily and form positively charged ions therefore metals are called electropositive elements. They have high densities (except Sodium and Potassium).They have high melting and boiling points (except Sodium and potassium). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
